ORIGINAL RESEARCH
Epithelial-Mesenchymal Transition-Like Immunophenotypic Changes in Gallbladder Carcinogenesis: A Quantitative Digital Pathology Study
ABSTRACT
Background: Gallbladder carcinoma develops through a multistep sequence from chronic inflammation to dysplasia and invasive carcinoma. The role of epithelial–mesenchymal transition (EMT)-associated phenotypic alterations in this process remains incompletely defined.
Objectives: To quantitatively evaluate immunohistochemical markers associated with EMT across the spectrum of gallbladder lesions and assess their relationship with disease progression.
Methods: A retrospective multicenter study was conducted on 90 formalin-fixed paraffin-embedded gallbladder specimens collected between 2015 and 2025, including chronic cholecystitis, intestinal metaplasia, biliary intraepithelial neoplasia (BilIN 1–3), and invasive adenocarcinoma. Immunohistochemical analysis was performed for E-cadherin, β-catenin, Vimentin, CK17, CK19, and Ki67. A quantitative assessment was conducted using digital whole-slide imaging (QuPath and ImageJ). An exploratory EMT-like index was calculated based on epithelial and mesenchymal marker expression. Statistical analysis was performed using nonparametric tests and correlation analysis.
Results: A progressive decrease in E-cadherin expression and a corresponding increase in Vimentin and nuclear β-catenin expression were observed across lesion categories (p<0.05). CK17 expression and the Ki-67 proliferative index were significantly elevated in dysplastic and malignant lesions compared with benign conditions (p<0.01). The EMT-like index showed a stepwise increase from inflammatory lesions to invasive carcinoma (p<0.01), indicating a shift toward a mesenchymal-associated immunophenotype during disease progression.
Conclusions: Gallbladder carcinogenesis is associated with progressive immunophenotypic changes characterized by reduced epithelial marker expression, increased mesenchymal marker expression, and enhanced proliferative activity. These findings are consistent with EMT-like phenotypic remodeling during tumor progression; however, mechanistic confirmation requires further molecular investigation.
Keywords: Gallbladder carcinoma; biliary intraepithelial neoplasia (BilIN 1–3); digital pathology; epithelial–mesenchymal transition; immunohistochemistry; tumor progression.
BACKGROUND
Gallbladder carcinoma represents the most common malignancy of the biliary tract and remains one of the most aggressive gastrointestinal tumors, characterized by late clinical presentation, rapid progression, and poor prognosis.1 Despite advances in surgical and oncological management, the overall survival rate for patients with gallbladder adenocarcinoma remains low, largely due to delayed diagnosis and early metastatic dissemination. Understanding the biological mechanisms underlying gallbladder carcinogenesis is therefore essential for improving diagnostic and prognostic stratification.
Gallbladder carcinogenesis is widely believed to follow a multistep sequence analogous to other epithelial malignancies, progressing from chronic inflammation through metaplastic and dysplastic changes to invasive carcinoma.2-4 Chronic cholecystitis, frequently associated with cholelithiasis, results in persistent epithelial injury and regeneration that may predispose to mucosal alterations, such as intestinal metaplasia.5 These metaplastic changes can subsequently progress to biliary intraepithelial neoplasia (BilIN), which is recognized as the dysplastic precursor of gallbladder carcinoma. BilIN lesions are classified into low-grade (BilIN-1), intermediate-grade (BilIN-2), and high-grade dysplasia (BilIN-3), reflecting increasing degrees of cytologic and architectural atypia.6,7 However, the molecular and phenotypic alterations driving the transition from benign inflammatory lesions to invasive carcinoma remain incompletely understood.
One of the key biological processes implicated in tumor progression and metastasis is epithelial–mesenchymal transition (EMT). EMT is a complex cellular program through which epithelial cells lose their characteristic polarity and cell-cell adhesion while acquiring mesenchymal properties that promote motility, invasion, and resistance to apoptosis.8,9 This process plays an important role in embryonic development and wound healing, but is increasingly recognized as a central mechanism in cancer progression. In carcinomas, EMT contributes to tumor invasion, dissemination, and the establishment of metastatic disease.10
At the molecular level, EMT is characterized by a shift in the expression of epithelial and mesenchymal markers. Loss of E-cadherin, a key epithelial adhesion molecule encoded by the CDH1 gene, is one of the most consistent features of EMT and results in reduced intercellular adhesion and increased tumor cell mobility.11 Tumor cells frequently acquire expression of mesenchymal cytoskeletal proteins such as Vimentin, which is associated with increased cellular plasticity and invasive potential. Alterations in the Wnt/β-catenin signaling pathway also play a crucial role in EMT activation. In epithelial tissues, β-catenin is normally localized to the cell membrane as part of adherens junctions; however, during EMT activation, it may accumulate in the cytoplasm and nucleus, where it functions as a transcriptional co-activator promoting tumor progression.11
In addition to classical EMT markers, other cytokeratins have been implicated in the malignant transformation of biliary epithelium. Cytokeratin 17 (CK17), a basal-type keratin associated with cellular stress and regenerative responses, has been increasingly recognized as a marker of tumor aggressiveness in several epithelial malignancies. In biliary tract tumors, CK17 expression has been linked to higher proliferative activity and more aggressive tumor behavior. Conversely, cytokeratin 19 (CK19), a well-established marker of biliary epithelial differentiation, is typically preserved across benign and malignant biliary lesions and therefore serves as a useful lineage marker in studies of gallbladder tumorigenesis.
The present study aimed to investigate the phenotypic features of epithelial–mesenchymal transition across different stages of gallbladder mucosal pathology. Using a retrospective multicenter cohort of gallbladder specimens, we performed immunohistochemical analysis of key EMT markers, including E-cadherin, Vimentin, and β-catenin, as well as additional markers of biliary differentiation and proliferative activity. Digital pathology tools were applied to quantify immunohistochemical expression patterns and to construct a composite EMT index reflecting the balance between epithelial and mesenchymal phenotypes. By comparing chronic cholecystitis, intestinal metaplasia, biliary intraepithelial neoplasia, and invasive adenocarcinoma, this study aims to clarify the role of EMT-related alterations in the stepwise progression of gallbladder carcinogenesis.
METHODS
Study design and case selection
This retrospective multicenter study was conducted at the Department of Molecular Pathology, Tbilisi State Medical University, which serves as a central diagnostic and research laboratory that receives surgical pathology specimens from multiple collaborating institutions in Tbilisi, Batumi, and Kutaisi. Archival formalin-fixed paraffin-embedded (FFPE) gallbladder tissue specimens obtained from cholecystectomy and tumor resection cases between 2015 and 2025 were retrieved from the institutional pathology archive. Cases were included based on the availability of adequate histological material and clinical data.
The study cohort comprised a spectrum of gallbladder lesions representing different stages of mucosal pathology, including chronic cholecystitis, intestinal metaplasia, biliary intraepithelial neoplasia (BilIN 1–3), and invasive gallbladder adenocarcinoma. In specimens with multiple lesion types, the highest-grade lesion was used for classification and statistical analysis. Cases were excluded if tissue preservation was inadequate, if extensive cautery artifacts were present, or if the diagnosis could not be confirmed upon histopathological review.
Histopathological evaluation
All hematoxylin and eosin–experienced pathologists independently reviewed stained slides to confirm the diagnosis and classify lesions according to established histopathological criteria. Chronic cholecystitis was defined by chronic inflammatory infiltration of the gallbladder wall accompanied by epithelial reactive changes. In contrast, intestinal metaplasia was identified by the presence of intestinal-type epithelium with goblet cells replacing native biliary epithelium. Dysplastic lesions were classified as biliary intraepithelial neoplasia (BilIN) according to the World Health Organization classification and categorized as low-grade (BilIN-1), intermediate-grade (BilIN-2), or high-grade (BilIN-3). Invasive carcinoma was diagnosed based on the presence of infiltrative malignant glandular structures invading the gallbladder wall. For carcinoma cases, additional clinicopathological parameters, including tumor differentiation grade, depth of invasion (pT stage), and lymphovascular invasion, were recorded.
Immunohistochemistry
Sections were cut at 4 μm from representative FFPE tissue blocks. Sections were mounted on positively charged glass slides and subjected to routine deparaffinization and rehydration. Staining was performed on an automated Leica Bond immunostaining platform (Leica Biosystems, Germany) using the Bond Polymer Refine Detection system, and heat-induced epitope retrieval was performed according to antibody-specific protocols. The primary antibodies used in this study included E-cadherin (clone NCH-38, 1:100, membranous localization), Vimentin (clone V9, 1:200, cytoplasmic localization), β-catenin (clone 14, 1:150, membranous and nuclear localization), CK17 (clone E3, 1:100, cytoplasmic localization), CK19 (clone A53-B/A2, 1:150, cytoplasmic localization), and Ki67 (clone MIB-1, 1:200, nuclear localization) (Tab.1).
TABLE 1. The primary antibodies with clones and dilutions

Abbreviations: A53-B/A2, mouse monoclonal antibody that targets cytokeratin 19; CK17, cytokeratin 17; CK19, cytokeratin 19; E3, antibodies against ApoE, Cytokeratin 17, or E3 ubiquitin ligases; Ki67, proliferation index; MIB-1, mouse monoclonal antibody (IgG1/kappa) against Ki-67; NCH-38, mouse monoclonal antibody (IgG1, kappa) against E-cadherin; V9, monoclonal antibody against vimentin; 14, refers to monoclonal antibodies against CD14, a glycoprotein expressed on monocytes and macrophages.
Immunohistochemical evaluation
Immunohistochemical expression was assessed by both conventional microscopic evaluation and digital image analysis. E-cadherin expression was assessed using a semi-quantitative H-score ranging from 0 to 300, incorporating both staining intensity and the proportion of positive epithelial cells. Vimentin and CK17 expression were quantified as the percentage of positively stained epithelial cells. β-catenin expression was evaluated with particular attention to nuclear localization, which was considered indicative of Wnt/β-catenin signaling pathway activation. The Ki67 proliferative index was calculated as the percentage of positively stained nuclei among at least 500 epithelial cells per case. CK19 expression was assessed as a lineage marker and quantified as the percentage of positive epithelial cells.
Digital pathology analysis
Whole-slide images were generated using a Motic digital slide scanner, and digital analysis was performed using QuPath software (version 0.4) and ImageJ. For each case, one representative tissue block was selected, and three to five regions of interest (ROIs) were manually annotated to include representative epithelial areas while excluding necrotic, cauterized, and stromal regions. Automated cell detection algorithms were applied to quantify staining intensity and the proportion of positive cells, with at least 500 epithelial cells analyzed per case. Positivity thresholds were defined based on DAB signal intensity calibrated against internal positive controls. Observers performing digital quantification were blinded to the diagnostic category, and a subset of cases was independently re-evaluated to assess reproducibility.
EMT-like index calculation
To evaluate the balance between epithelial and mesenchymal phenotypes, a composite EMT-like index was calculated for each case using the formula: (Vimentin expression percentage + nuclear β-catenin expression percentage) divided by the E-cadherin H-score. This index was used as an exploratory indicator of EMT-like phenotypic shift, reflecting the relative loss of epithelial characteristics and acquisition of mesenchymal features.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics (version 26). Continuous variables were expressed as mean ± standard deviation or median ± interquartile range, depending on the data distribution. Comparisons between lesion groups were performed using nonparametric tests, including the Kruskal–Walli’s test for multiple-group comparisons and the Mann–Whitney U test for pairwise comparisons. Correlations between variables were assessed using Spearman’s rank correlation coefficient, and a p-value of less than 0.05 was considered statistically significant.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki. Ethical approval was obtained from the Ethics Committee of Tbilisi State Medical University (Approval No: TSMU-EC/2021/05-20), and all patient data were anonymized prior to analysis.
RESULTS
Clinicopathological characteristics of the study cohort
A total of 90 gallbladder specimens representing inflammatory, pre-neoplastic, and neoplastic lesions were included in the study. The cohort comprised cases of chronic cholecystitis, intestinal metaplasia, biliary intraepithelial neoplasia (BilIN 1–3), and invasive gallbladder adenocarcinoma (Tab.2).
TABLE 2. Clinicopathological characteristics of the study cohort

Abbreviations: BilIN-1, low-grade biliary intraepithelial neoplasia; BilIN-2, intermediate-grade biliary intraepithelial neoplasia; BilIN-3, high-grade biliary intraepithelial neoplasia; CIS, carcinoma in situ; F, female; M, male; SD, standard deviation.
Overall, patients ranged in age from approximately 34 to 79 years, with a slight predominance of female patients, consistent with the known epidemiological pattern of gallbladder disease. Carcinoma cases demonstrated variable histological differentiation and were further evaluated for depth of invasion and lymphovascular invasion.
Epithelial marker expression across the lesion spectrum
E-cadherin expression decreased progressively across the spectrum of gallbladder lesions. In chronic cholecystitis and intestinal metaplasia, strong membranous staining was observed in the majority of epithelial cells, corresponding to high H-scores. In contrast, dysplastic lesions showed a gradual reduction in membranous expression, with BilIN-3 lesions demonstrating marked attenuation. In invasive adenocarcinoma, E-cadherin expression was frequently reduced or heterogeneous, with partial or complete loss of membranous staining in tumor glands. Quantitative analysis confirmed a statistically significant decrease in E-cadherin expression across lesion categories (p<0.001), supporting progressive loss of epithelial adhesion during disease progression.
CK19 expression remained consistently high across all lesion categories, without statistically significant variation, confirming its role as a stable marker of biliary epithelial differentiation rather than a dynamic indicator of malignant transformation.
Mesenchymal marker expression and EMT-like changes
Vimentin expression was absent or minimal in epithelial cells of chronic cholecystitis and intestinal metaplasia (Tab.3). However, focal epithelial vimentin positivity emerged in dysplastic lesions and became increasingly prominent in invasive adenocarcinoma. A similar pattern was observed for β-catenin localization. While benign lesions predominantly exhibited membranous β-catenin staining, dysplastic and malignant lesions demonstrated progressive nuclear accumulation. Quantitative analysis using the Kruskal-Wallis test revealed a significant increase in both vimentin expression and nuclear β-catenin positivity across lesion groups (p<0.001), indicating a shift toward a mesenchymal-associated immunophenotype during gallbladder carcinogenesis (Tab.3).
TABLE 3. Quantitative immunohistochemical marker expression across gallbladder lesions
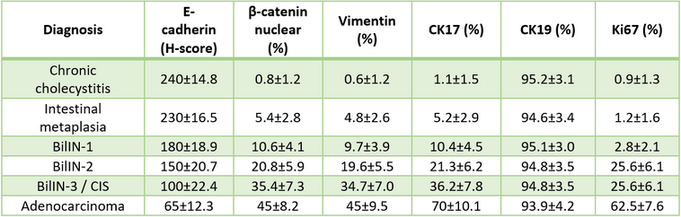
Abbreviations: BilIN-1, low-grade biliary intraepithelial neoplasia; BilIN-2, intermediate-grade biliary intraepithelial neoplasia; BilIN-3, high-grade biliary intraepithelial neoplasia; CK17, cytokeratin 17; CK19, cytokeratin 19; H-score, histological score; Ki67, proliferation index; SD, standard deviation. Values are presented as mean ± standard deviation.
CK17 expression and proliferative activity
CK17 expression was minimal or absent in chronic cholecystitis and intestinal metaplasia. In contrast, CK17 positivity increased progressively in dysplastic lesions and was most pronounced in invasive adenocarcinoma, where a substantial proportion of tumor cells exhibited cytoplasmic staining. This progressive increase in CK17 expression suggests its association with epithelial transformation and tumor progression (Fig.1).
FIGURE 1. The gallbladder adenocarcinoma whole-slide imaging analysis

Explanations: A. Whole-slide image (WSI) of gallbladder adenocarcinoma; B. Higher magnification showing CK17 immunohistochemical staining; C. High-power view illustrating heterogeneous CK17 expression among tumor glands.
The Ki67 proliferative index demonstrated a similar trend, with low proliferative activity in benign lesions and a marked increase in dysplastic and malignant conditions. Chronic inflammatory lesions exhibited Ki67 positivity, largely confined to the basal epithelial layers, whereas invasive carcinoma showed diffuse, high proliferative indices. Statistical analysis confirmed a significant increase in Ki67 expression across the lesion spectrum (p<0.001).
EMT-like index across gallbladder lesions
To quantitatively assess the balance between epithelial and mesenchymal phenotypes, a composite EMT-like index was calculated for each case. The EMT-like index showed a stepwise increase across lesion categories, with the lowest values in chronic cholecystitis and intestinal metaplasia, intermediate values in BilIN lesions, and the highest in invasive adenocarcinoma. This trend was statistically significant (p<0.001) and reflects a progressive shift toward a mesenchymal-associated immunophenotype during gallbladder carcinogenesis.
Correlation with Clinicopathological Parameters: In invasive carcinoma cases, higher EMT-like index values were associated with adverse pathological features. Specifically, increased EMT-like index correlated positively with higher tumor grade and the presence of lymphovascular invasion, suggesting a relationship between EMT-associated phenotypic changes and tumor aggressiveness. These findings support the potential relevance of EMT-related marker expression in evaluating gallbladder carcinoma progression (Fig.2).
FIGURE 2. EMT-related marker expression in gallbladder carcinoma progression

Explanations: A. EMT-like index across gallbladder lesion categories; B. E-cadherin expression (H-score); C. CK17 expression across lesion categories; D. Vimentin expression.
DISCUSSION
Gallbladder carcinoma represents a highly aggressive malignancy characterized by late clinical presentation and poor prognosis. Chronic inflammatory conditions of the gallbladder, particularly cholelithiasis-associated chronic cholecystitis, are recognized as important risk factors in the development of gallbladder carcinoma. The transition from chronic inflammation to metaplastic and dysplastic epithelial alterations has been described as a potential pathway leading to malignant transformation in the gallbladder mucosa.
In the present study, we investigated the expression of epithelial and mesenchymal-associated markers across a spectrum of gallbladder lesions, including inflammatory, pre-neoplastic, and malignant conditions. The results demonstrated a gradual reduction in epithelial marker expression, particularly E-cadherin, accompanied by increased expression of mesenchymal markers, including Vimentin and nuclear β-catenin. These findings indicate a progressive immunophenotypic shift during gallbladder carcinogenesis. However, these alterations should be interpreted as an EMT-like pattern rather than definitive evidence of epithelial–mesenchymal transition as a biological process, as immunohistochemical analysis reflects static protein expression rather than dynamic molecular mechanisms.
Loss of E-cadherin-mediated cell adhesion is considered a hallmark feature of EMT.12 Reduced E-cadherin expression has been described in multiple epithelial malignancies, including gallbladder carcinoma, where it is associated with increased invasiveness and metastatic potential.13 In our cohort, E-cadherin expression was preserved in benign inflammatory lesions but progressively decreased in dysplastic and malignant epithelium, suggesting that disruption of epithelial cell adhesion represents an early phenotypic alteration in gallbladder carcinogenesis. Nevertheless, in the absence of molecular validation, this finding should be interpreted as part of a broader process of epithelial remodeling rather than direct confirmation of EMT activation.
The increased expression of Vimentin observed in dysplastic and malignant lesions further supports the acquisition of mesenchymal-associated features during tumor progression. Vimentin expression in epithelial tumors is commonly associated with enhanced migratory capacity and tumor aggressiveness.14 Similarly, analysis of β-catenin localization demonstrated a progressive shift from membranous expression in benign lesions to nuclear accumulation in dysplastic and malignant epithelium.15,16 Nuclear β-catenin participates in Wnt signaling pathways involved in tumor proliferation and invasion.17 The combined increase in Vimentin expression and nuclear β-catenin localization suggests a coordinated shift toward a mesenchymal-associated phenotype; however, these findings should be interpreted cautiously, as immunohistochemical evidence alone does not establish mechanistic pathway activation.
Another noteworthy finding of the present study was the progressive increase in CK17 expression across dysplastic and malignant lesions. CK17 has been described as a marker of epithelial proliferation and tumor aggressiveness in several carcinomas, including biliary tract malignancies.18 The observed upregulation of CK17 may reflect alterations in epithelial differentiation and stress-response pathways, suggesting that its expression is associated with phenotypic adaptation during tumor progression rather than representing a purely secondary phenomenon.
The Ki67 proliferative index also demonstrated a clear increase across lesion categories, supporting the concept that proliferative activity increases during the transition from benign to malignant gallbladder lesions. The concurrent increase in proliferative activity alongside the emergence of mesenchymal-associated markers suggests a potential relationship between epithelial plasticity and tumor growth dynamics. This observation is consistent with the concept that increased proliferation and loss of epithelial characteristics may occur simultaneously during tumor progression, contributing to a more aggressive biological phenotype.
The introduction of a composite EMT-like index in this study provides an integrative approach to evaluating the balance between epithelial and mesenchymal features. Although this index represents an exploratory metric rather than a validated scoring system, its progressive increase across lesion categories supports its potential utility as a quantitative indicator of phenotypic transition. Further studies incorporating molecular validation and larger patient cohorts are required to determine its reproducibility and clinical significance.
Taken together, the present findings suggest that gallbladder carcinogenesis is associated with a progressive shift in immunophenotypic characteristics, involving loss of epithelial adhesion, acquisition of mesenchymal-associated features, and increased proliferative activity. These changes appear to occur in a stepwise manner across the spectrum from benign inflammatory conditions to invasive carcinoma, supporting the concept of gradual phenotypic remodeling during tumor development.
From a clinical perspective, the identification of progressive EMT-like immunophenotypic changes across gallbladder lesions may have potential implications for risk stratification and prognostic assessment. Markers such as CK17, Ki67, and the composite EMT-like index can help identify lesions with a higher potential for progression. However, further validation in clinically annotated cohorts is necessary before routine application.
Certain clinicopathological distributions observed in the present cohort may reflect sampling characteristics inherent to retrospective multicenter studies; therefore, caution should be exercised when generalizing these findings. In addition, the retrospective nature of the study and the multicenter origin of the specimens may introduce variability in tissue processing and clinical data. Molecular genetic analyses were not performed, which limits the ability to define the mechanistic basis of the observed phenotypic changes. Future studies integrating molecular and transcriptomic approaches are needed to further elucidate the biological pathways underlying gallbladder tumor progression.
CONCLUSIONS
The present study demonstrates a progressive shift in immunophenotypic characteristics across the spectrum of gallbladder lesions, characterized by decreased epithelial marker expression, increased mesenchymal-associated marker expression, and enhanced proliferative activity. These findings are consistent with an EMT-like pattern of phenotypic remodeling during gallbladder carcinogenesis.
The observed increase in CK17 expression and Ki67 proliferative index, together with alterations in E-cadherin, Vimentin, and β-catenin expression, suggests that coordinated changes in differentiation, proliferation, and cellular plasticity accompany epithelial transformation. The composite EMT-like index introduced in this study provides a quantitative approach to assessing this phenotypic transition, although further validation is required.
While these results highlight potential markers associated with disease progression, the findings should be interpreted cautiously, as immunohistochemical analysis alone does not establish the mechanistic basis of epithelial-mesenchymal transition. Future studies incorporating molecular and functional analyses, as well as larger and clinically annotated cohorts, are necessary to clarify the biological significance and potential clinical utility of these observations.
AUTHOR AFFILIATION
1Department of Molecular Pathology, Faculty of Medicine, Tbilisi State Medical University, Tbilisi, Georgia;
2Diagnostic Research and Scientific Laboratory, Tbilisi State Medical University, Tbilisi, Georgia.
REFERENCES
-
Roa JC, Basturk O, Adsay V. Dysplasia and carcinoma of the gallbladder: pathological evaluation, sampling, differential diagnosis and clinical implications. Histopathology. 2021 Jul 1;79(1):2–19. doi:10.1111/his.14360 PubMed PMID: 33629395.
-
Roa JC, García P, Kapoor VK, Maithel SK, Javle M, Koshiol J. Gallbladder cancer. Nat Rev Dis Primers. 2022 Dec 1;8(1):69. doi:10.1038/s41572-022-00398-y PubMed PMID: 36302789.
-
Goetze TO. Gallbladder carcinoma: Prognostic factors and therapeutic options. World J Gastroenterol. 2015 Nov 7;21(43):12211–12217. doi:10.3748/wjg.v21.i43.12211. PMID: 26556987.
-
Hundal R, Shaffer EA. Gallbladder cancer: Epidemiology and outcome. Clin Epidemiol. 2014 Mar 7;6(1):99–109. doi:10.2147/CLEP.S37357 PubMed PMID: 24634588.
-
Seretis C, Lagoudianakis E, Salemis NS, Seretis F. Metaplastic changes in chronic cholecystitis: implications for early diagnosis and surgical intervention. J Clin Med Res. 2013;5(5):343–347. doi:10.4021/jocmr1689w.
-
Bridgewater JA, Goodman KA, Kalyan A, Mulcahy MF. Biliary Tract Cancer: Epidemiology, Radiotherapy, and Molecular Profiling. American Society of Clinical Oncology Educational Book. 2016 May;(36):e194–203. doi:10.1200/edbk_160831 PubMed PMID: 27249723.
-
Miranda-Filho A, Piñeros M, Ferreccio C, Adsay V, Soerjomataram I, Bray F, et al. Gallbladder and extrahepatic bile duct cancers in the Americas: Incidence and mortality patterns and trends. Int J Cancer. 2020 Aug 15;147(4):978–89. doi:10.1002/ijc.32863 PubMed PMID: 31922259.
-
Puisieux A, Brabletz T, Caramel J. Oncogenic roles of EMT-inducing transcription factors. Nat Cell Biol. 2014;16(6):488–94. doi:10.1038/ncb2976 PubMed PMID: 24875735.
-
Sánchez-Tilló E, Liu Y, De Barrios O, Siles L, Fanlo L, Cuatrecasas M, et al. EMT-activating transcription factors in cancer: Beyond EMT and tumor invasiveness. Cellular and Molecular Life Sciences. 2012 Oct;69(20):3429–56. doi:10.1007/s00018-012-1122-2 PubMed PMID: 22945800.
-
Tiwari N, Gheldof A, Tatari M, Christofori G. EMT as the ultimate survival mechanism of cancer cells. Semin Cancer Biol. 2012 Jun;22(3):194–207. doi:10.1016/j.semcancer.2012.02.013 PubMed PMID: 22406545.
-
Craene B De, Berx G. Regulatory networks defining EMT during cancer initiation and progression. Nat Rev Cancer. 2013 Feb;13(2):97–110. doi:10.1038/nrc3447 PubMed PMID: 23344542.
-
Heuberger J, Birchmeier W. Interplay of cadherin-mediated cell adhesion and canonical Wnt signaling. Cold Spring Harb Perspect Biol. 2010;2(2). doi:10.1101/cshperspect.a002915 PubMed PMID: 20182623.
-
Roa JC, Roa I, Correa P, Vo Q, Araya JC, Villaseca M, et al. Microsatellite instability in preneoplastic and neoplastic lesions of the gallbladder. J Gastroenterol. 2005 Jan;40(1):79–86. doi:10.1007/s00535-004-1497-4 PubMed PMID: 15692793.
-
Thiery JP, Acloque H, Huang RYJ, Nieto MA. Epithelial-Mesenchymal Transitions in Development and Disease. Cell. 2009 Nov 25;139(5):871–90. doi:10.1016/j.cell.2009.11.007 PubMed PMID: 19945376.
-
Nusse R, Fuerer C, Ching W, Harnish K, Logan C, Zeng A, et al. Wnt signaling and stem cell control. Cold Spring Harb Symp Quant Biol. 2008;73:59–66. doi:10.1101/sqb.2008.73.035 PubMed PMID: 19028988.
-
Zhang J, Tian XJ, Xing J. Signal transduction pathways of EMT induced by TGF-β, SHH, and WNT and their crosstalks. J Clin Med. 2016 Apr 1;5(4). doi:10.3390/jcm5040041
-
Chang HJ, Jee C Do, Kim WH. Mutation and altered expression of β-catenin during gallbladder carcinogenesis. American Journal of Surgical Pathology. 2002;26(6):758–66. doi:10.1097/00000478-200206000-00009 PubMed PMID: 12023580.
-
Matsushita S, Onishi H, Nakano K, Nagamatsu I, Imaizumi A, Hattori M, et al. Hedgehog signaling pathway is a potential therapeutic target for gallbladder cancer. Cancer Sci. 2014;105(3):272–80. doi:10.1111/cas.12354 PubMed PMID: 24438533.
