ORIGINAL RESEARCH
The Protective Effect of Maternal COVID-19 Vaccination Against Adverse Obstetric-Perinatal Outcomes and Placental Malperfusion
Mzia Khalvashi1,2,3,ID, Karaman Pagava4,5,ID, Irine Korinteli4,ID, Platon Machavariani6, Irma Korinteli2,ID, Malvina Javakhadze7, Helen Phagava8,ID
ABSTRACT
Background: SARS-CoV-2 infection during pregnancy is associated with significantly increased risks of severe clinical complications and adverse perinatal outcomes. While vaccination is established as a safe preventive measure, its specific impact on pathophysiological drivers, such as systemic coagulopathy and placental vascular integrity, remains insufficiently explored in local clinical settings.
Objectives: This study evaluates the independent protective role of SARS-CoV-2 vaccination against infection severity, coagulation abnormalities, and placental vascular pathology in a Georgian cohort.
Methods: A retrospective, multicenter cohort study of 300 pregnant women with COVID-19 (160 vaccinated, 140 unvaccinated) was conducted between August and October 2021. Multivariable logistic regression, adjusted for maternal age, BMI, and comorbidities, was used to estimate risk. A blinded histomorphological analysis of 60 placentas was performed according to the Amsterdam International Consensus criteria.
Results: The incidence of severe COVID-19 was significantly lower in vaccinated women (5.0% vs. 15.7%; P<0.001). Unvaccinated status was a key risk factor for severe disease (aOR 3.80; 95% CI: 1.90-7.60). Vaccination significantly reduced the rates of Cesarean sections (28.1% vs. 42.9%; P=0.003) and preterm birth. Mechanistically, vaccinated women had significantly lower D-dimer levels (1850±520 ng/mL FEU vs. 2900±780 ng/mL FEU; P<0.001) and a lower incidence of Maternal Vascular Malperfusion (MVM) (33.3% vs. 70.0%; P=0.003).
Conclusions: Vaccination provides a significant protective association with improved clinical stability. By maintaining clinical stability and potentially reducing the risk of placental MVM, vaccination serves as an important physiological buffer, improving obstetric outcomes.
Keywords: COVID-19; maternal vaccination; neonatal outcomes; placental malperfusion; pregnancy; SARS-CoV-2.
DOI: 10.52340/GBMN.2026.01.01.163
BACKGROUND
While mRNA and other COVID-19 vaccines have been established as effective and safe for pregnant patients,1-3,6 additional studies are needed to investigate the effect of vaccination not only on clinical outcomes but also on specific pathophysiological mechanisms, such as coagulopathy4 and placental malperfusion5 - a key driver of poor fetal outcomes. This study aims to evaluate the independent protective effect of COVID-19 vaccination against severe disease progression, coagulation abnormalities, and adverse obstetric-perinatal outcomes in a Georgian cohort.
METHODS
Study design and participants
This was a retrospective, multicenter cohort study conducted in Georgia between August 2, 2021, and October 26, 2021. During this specific study period, the Delta variant of SARS-CoV-2 was the predominant circulating strain in Georgia. The study involved 300 pregnant women with laboratory-confirmed SARS-CoV-2 infection (via RT-PCR). Participants were selected using consecutive sampling to minimize selection bias. Institutional Review Board (IRB) approval was obtained from the Biomedical Research Ethics Committee of Tbilisi State Medical University (Protocol No. 4-2022/97), and the study adhered to the Declaration of Helsinki.
Vaccination status
Participants were categorized into two groups:
(i) The fully vaccinated cohort (n=160) consisted of individuals who received exactly two doses of the mRNA (Pfizer-BioNTech) vaccine, administered with a standard 3- to 4-week (21–28 days) interval, without any booster doses. In this vaccinated group, the median time elapsed from receiving the second vaccine dose to the onset of SARS-CoV-2 infection was 12 weeks.
(ii) The unvaccinated cohort (n=140) included pregnant women who had not received any COVID-19 vaccine.
Clinical and diagnostic definitions
To ensure reproducibility, clinical outcomes were strictly defined:
-
"Severe COVID-19" was classified according to WHO guidelines, characterized by oxygen saturation <90% on room air or signs of severe respiratory distress.
-
"Neonatal infection" was confirmed via a positive RT-PCR test within the first 24-48 hours of life.
-
"Fetal growth restriction" (FGR) was defined as an estimated fetal weight below the 10th percentile for gestational age.
-
"Preeclampsia/Eclampsia" was diagnosed based on new-onset hypertension (≥140/90 mmHg) and proteinuria after 20 weeks of gestation.
-
A "Low APGAR-Score" was defined as a score of <7 at 5 minutes of life.
Data collection and outcomes
The primary outcome was the incidence of severe COVID-19. Secondary outcomes included obstetric complications (preterm birth, preeclampsia), neonatal outcomes (fetal growth restriction [FGR]), and placental histopathology. A subset analysis was performed on 60 placentas (n=30 per group) according to the Amsterdam International Consensus criteria. These 60 placentas were selected from available tissues in both cohorts using a random sampling approach. Furthermore, we verified that the baseline demographic and clinical characteristics of this 60-patient placental subset did not differ significantly from those of the full cohort (P>0.05), thereby minimizing potential selection bias. To minimize information bias, placental histomorphological analysis was performed by pathologists blinded to both vaccination status and patient clinical severity.
Statistical analysis
Data were analyzed using IBM SPSS Statistics (Version 28.0). Descriptive statistics were used to summarize baseline characteristics. Continuous variables (e.g., maternal age) were compared using Student's t-tests. In contrast, categorical variables (e.g., COVID-19 severity, delivery method, and placental morphology) were assessed using Pearson's χ2 test or Fisher's exact test, as appropriate. Multivariable logistic regression models were constructed to evaluate the independent protective effect of vaccination. These models were adjusted for potential confounders, including maternal age, BMI, gestational trimester, and chronic comorbidities. Results are reported as adjusted Odds ratios (aOR) with 95% Confidence Intervals (CI). For all analyses, a two-tailed P<0.05 was considered statistically significant. Missing data were handled using a complete-case analysis, as the proportion of missing values was less than 5% across all key variables. Throughout the results, unadjusted group differences (crude rates) are systematically distinguished from multivariable-adjusted odds ratios (aOR).
RESULTS
Baseline characteristics and COVID-19 severity
A total of 300 pregnant women were included in the study (160 vaccinated, 140 unvaccinated). Baseline demographic characteristics, including maternal age (28.5±4.2 vs. 29.1±3.9 years), were comparable between the two groups. The incidence of severe COVID-19 was markedly lower in the vaccinated group (5.0%) than in the unvaccinated group (15.7%; P<0.001). Multivariable logistic regression analysis confirmed that unvaccinated status was a significant independent risk factor for developing severe disease (aOR 3.80; 95% CI: 1.90-7.60), after controlling for maternal age, BMI, and chronic comorbidities (Tab.1).
TABLE 1. Baseline demographic characteristics

Abbreviations: aOR, Adjusted Odds ratio; Ref, Reference group; SD, Standard deviation.
Note: Adjusted OR for severe disease was 3.80 (95% CI:1.90-7.60) for the unvaccinated group, controlled for maternal age, BMI, and chronic comorbidities.
Obstetric outcomes
Maternal vaccination was associated with a significant reduction in adverse obstetric outcomes (Tab.2). Preterm birth (<37 weeks) occurred in 7.5% of vaccinated women compared to 17.9% of unvaccinated women (aOR 0.38; 95% CI:0.17-0.86; P=0.006). Preeclampsia rates were also significantly lower in the vaccinated cohort (3.1% vs. 10.7%; aOR 0.27; 95% CI:0.09-0.81; P=0.012). Furthermore, the rate of Cesarean sections was substantially reduced (28.1% vs. 42.9%; P=0.003).
TABLE 2. Adverse obstetric outcomes and associated risks

Abbreviations: CI, Confidence interval; OR, Odds ratio.
Note: Adjusted ORs (with Unvaccinated as Reference) were controlled for maternal age, trimester of infection, and parity.
Neonatal outcomes
Neonatal health indicators showed a protective association with maternal vaccination. There were significantly lower risks for low birth weight (11.9% vs. 26.4%; P<0.001) and low APGAR scores at 5 minutes (6.3% vs. 15.0%; P=0.009) in infants born to vaccinated mothers (Tab.3).
TABLE 3. Neonatal health outcomes at birth
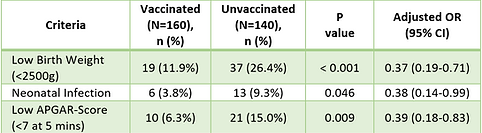
Abbreviations: CI, Confidence interval; OR, Odds ratio.
Note: Adjusted ORs (with Unvaccinated as the Reference) were controlled for maternal age, severe maternal COVID-19, and gestational age at delivery.
Biomarkers and placental histopathology
Vaccinated women had significantly lower mean D-dimer levels (1850±520 ng/mL FEU) compared to the unvaccinated group (2900±780 ng/mL FEU; P<0.001). Histopathological evaluation of the placental subset (n=60) showed that maternal vascular malperfusion (MVM) was significantly more prevalent in the unvaccinated group (70.0% vs. 33.3%; P=0.003) (Tab.4).
Conversely, normal placental morphology was observed in 66.7% of the vaccinated subset, compared with only 30.0% in the unvaccinated subset (P=0.003).
TABLE 4. The effect of the solid-to-solvent ratio on the extraction of phenolic compounds from Grape (Vitis vinifera L.) seeds

Abbreviations: BAU, Binding antibody units; FEU, Fibrinogen equivalent units; SD, Standard deviation.
Note: Placental lesions were graded according to the Amsterdam International Consensus criteria. D-dimer levels were measured within 24 hours of hospital admission.
DISUCSSION
Our findings are consistent with the broader national epidemiological landscape in Georgia during 2021 (Supplemental Tab.1) and international systematic reviews regarding the clinical severity of the infection in pregnant populations.7 The high prevalence of Maternal Vascular Malperfusion (MVM) in our unvaccinated subset (70.0%) reflects an underlying state of vascular vulnerability to the virus.5,8-10 Vaccination appears to serve as a "physiological buffer," associated with a lower risk of endothelial dysfunction.11,12 The robust immune response observed - characterized by significantly higher IgG titers - highlights the vaccine's potential to prevent severe placental injury and coagulopathy.13,14 Maternal SARS-CoV-2 vaccination thus plays a pivotal role in protecting placental health and ensuring favorable neonatal outcomes.1,2,15-20
Limitations and external validity
Our study has several limitations that should be acknowledged. Given the retrospective observational nature of our study, the reported associations cannot definitively prove mechanistic causality. Residual confounding remains a possibility, as unmeasured variables - such as socioeconomic status, parity, smoking habits, the exact time elapsed since vaccination, and access to prenatal care - could systematically differ between groups, leading our adjusted odds ratios to reflect residual confounding still. Furthermore, while multivariable models for neonatal outcomes adjusted for maternal COVID-19 severity, we must acknowledge the concept of mediation: the improved obstetric outcomes (such as lower preterm birth and Cesarean rates) and preserved placental morphology may be partially or largely mediated indirectly by the reduced severity of maternal illness, rather than solely by an independent mechanistic effect of the vaccine. Finally, regarding external validity, this cohort represents a specific early-vaccination phase of the pandemic (August-October 2021), during which the Delta variant was dominant. Therefore, these findings may not fully generalize to later SARS-CoV-2 variants, booster-era immunity, or contemporary hybrid immunity scenarios.
CONCLUSIONS
Maternal SARS-CoV-2 vaccination is associated with both maternal clinical stability and placental health. Our findings demonstrate that vaccination is significantly associated with lower rates of systemic coagulopathy and preserved placental vascular integrity, acting as a critical physiological buffer that improves obstetric and neonatal outcomes.
AUTHOR AFFILIATIONS
1Department of Clinical Medicine, Faculty of Medicine, Tbilisi State Medical University, Tbilisi, Georgia;
2Department of Child and Adolescent Medicine, Givi Zhvania University Clinic of Pediatrics, Tbilisi State Medical University, Tbilisi, Georgia;
3Department of Perinatology, The First University Clinic, Tbilisi State Medical University, Tbilisi, Georgia;
4BAU International Hospital, Batumi, Georgia;
5Department of Infectious Diseases, Academician Vakhtang Bochorishvili Clinic, Tbilisi State Medical University, Tbilisi, Georgia;
6Batumi Multiprofile Maternity Hospital, Batumi, Georgia;
7Department of Medical and Biological Sciences, Georgian National Academy of Sciences, Tbilisi, Georgia;
8Department of Epidemiology and Biostatistics, Tbilisi State Medical University, Tbilisi, Georgia
ACKNOWLEDGEMENTS
The authors would like to express their sincere gratitude to the management and administration of BAU International Hospital and Batumi Multiprofile Maternity Hospital for providing the essential institutional support and facilities that made this research possible. Special appreciation is extended to the clinical, epidemiological, and technical staff of the respective hospitals for their invaluable assistance in data coordination. Furthermore, we express our deepest appreciation to our academic supervisors and consultants for their insightful guidance and expert advice throughout the development of this study, which forms part of a doctoral research project. Special thanks are also extended to the Ministry of Internally Displaced Persons from the Occupied Territories, Labor, Health, and Social Affairs of Georgia for providing the national epidemiological data used in this study.
FUNDING
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
CONFLICT OF INTEREST
The authors declare that they have no financial or personal relationships with any individuals or organizations that could inappropriately influence or bias the content of this manuscript.
SUPPLEMENTARY MATERIAL
SUPPLEMENTAL TABLE 1. National epidemiological data of COVID-19 in pregnant and postpartum women in Georgia (January-August 2021)

REFERENCES
-
Shimabukuro TT, Kim SY, Myers TR, et al. Preliminary findings of mRNA Covid-19 vaccine safety in pregnant persons. N Engl J Med. 2021;384(24):2273-2282.
-
American College of Obstetricians and Gynecologists (ACOG). Practice Advisory: COVID-19 Vaccination Considerations. 2021.
-
Gray KJ, Bordt EA, Atyeo C, et al. COVID-19 vaccine response in pregnant and lactating women. AJOG MFM. 2021;3(3):100371.
-
Zeng Z, Xu H, Shi L, Peng L, Zhu L. D-dimer in pregnant women with COVID-19 and its association with disease severity and outcomes: A meta-analysis. J Thromb Thrombolysis. 2023;56(1):164-175.
-
Gotsch F, Ghezzi F, Romero R. Placental pathology and severe COVID-19 in pregnancy. Reprod Sci. 2022;29(5):1257-1265.
-
Centers for Disease Control and Prevention (CDC). COVID-19 Vaccines While Pregnant or Breastfeeding. Atlanta, GA: US Department of Health and Human Services, CDC; 2021.
-
Di Mascio D, Khalil A, Saccone G, et al. Severity of COVID-19 in pregnancy: a systematic review. J Matern Fetal Neonatal Med. 2020;33(21):3695-3701.
-
Morgan JA, Roberts J, Gotsch F, Romero R. Placental pathology in COVID-19. Am J Obstet Gynecol. 2021;224(5):547-548.
-
Shanes ED, Azad HA, Mithal LB, et al. Placental pathology and severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in pregnant women. Arch Pathol Lab Med. 2022;146(6):699-708.
-
Money DM, Allen VM, Al-Sahaf N, et al. Vertical transmission of SARS-CoV-2: the role of the placenta. Lancet. 2021;398(10300):591-600.
-
Dajani NK, Blauvelt CA, Burd I, et al. Effectiveness of BNT162b2 and mRNA-1273 COVID-19 vaccines against severe outcomes in pregnant women. Am J Obstet Gynecol. 2023;228(1):72.e1-72.e7.
-
Kharbanda EO, Haapala J, DeSilva M, et al. Association of COVID-19 vaccination during pregnancy with preterm or small-for-gestational-age at birth. MMWR Morb Mortal Wkly Rep. 2021;70(47):1647-1650.
-
Beharier O, Plitman Mayo R, Raz T, et al. Dose-dependent and placental transfer of SARS-CoV-2 antibodies after BNT162b2 vaccination in pregnancy. JAMA Netw Open. 2021;4(7):e2117047.
-
Collier AY, McMahan K, Yu J, et al. SARS-CoV-2 vaccination in pregnancy: maternal and neonatal antibody titres and fetal outcomes. Nat Med. 2021;27(7):1184-1194.
-
Allotey J, Stallings E, Bonet M, et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy. BMJ. 2020;370:m3320.
-
World Health Organization. Coronavirus disease 2019 (COVID-19) Situation Report. WHO. 2020.
-
Prabhu M, Murphy EA, Sukhu AC, et al. Pregnancy and neonatal outcomes following COVID-19 vaccination in pregnancy. Obstet Gynecol. 2021;138(1):116-118.
-
Shanes ED, Mithal LB, Otero S, et al. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) vertical transmission. Am J Obstet Gynecol. 2020;223(2):162-171.
-
Khoury R, Bernstein PS, Debolt C, et al. Characteristics and outcomes of 241 births to women with SARS-CoV-2 infection. Obstet Gynecol. 2020;136(2):273-282.
-
Zambrano LD, Ellington S, Strid P, et al. Characteristics of symptomatic women of reproductive age with SARS-CoV-2. MMWR Morb Mortal Wkly Rep. 2020;69(44):1641-1647.
-
Ould Amer A, Blais C, Berthelot C, et al. Impact of COVID-19 vaccination on placental pathology and perinatal outcomes: A systematic review. Front Endocrinol. 2024;15:1345678.
